Abstract
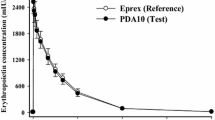
The purpose of this paper is: (i) to compare recombinant human erythropoietin (rHuEPO) pharmacokinetics in athletes and healthy individuals; and (ii) to report pharmacokinetic/pharmacodynamic (PK/PD) studies performed in athletes. Effect parameters in PK/PD studies included: (i) red blood cell variables (haematocrit, reticulocyte count); and (ii) markers of iron metabolism (serum soluble transferrin receptors [sTfR], ferritin [fr] and sTfR: fr ratio). To understand the choice of these markers, we first performed a brief review of the pharmacological effects of rHuEPO. Few studies have been conducted in healthy individuals and there are minimal references concerning pharmacokinetics in athletes. A ‘flip-flop’ phenomenon was noted after subcutaneous administration. The pharmacokinetics appeared linear from 50–1000 U/kg, but this linearity was not observed at the lowest dose of 10 U/kg. A negative-feedback loop of endogenous erythropoietin production occurred at the end of treatment. The half-life of the terminal part of the curves seemed to be slightly higher in athletes (36–42 vs 32 hours) than in untrained individuals and total clearance tended to be greater (17.5 vs 6.5 mL/h/ kg).
In conclusion, more investigations are needed to better understand the relationship between rHuEPO administration and changes in haematological and iron-metabolism parameters in athletes, particularly after chronic low-dose administration of rHuEPO.









Similar content being viewed by others
Notes
Use of tradenames is for product identification purposes only and does not imply endorsement.
References
Wide L, Bengtsson C, Birgegard G. Orcadian rhythm of erythropoietin in human serum. Br J Haematol 1989; 72: 85–90
Gareau R, Gagnon MG, Ayotte C, et al. rHuEpo increases urinary excretion of fibrin degradation products in haemodialysis patients. Thromb Haemost 1993; 70: 373–4
Wide L, Bengtsson C, Berglund B, et al. Detection in blood and urine of recombinant erythropoietin administered to healthy men. Med Sci Sports Exerc 1995; 27: 1569–76
Rivier R, Saugy M. Peptide hormones abuse in sport: state of the art in the detection of growth hormone and erythropoietin. J Toxicol Toxin Rev 1999; 18: 145–76
Lasne F, De Ceaurriz J. Recombinant erythropoietin in urine [letter]. Nature 2000 Jun; 405(6787): 635
Lasne F. Double-blotting: a solution to the problem of nonspecific binding of secondary antibodies in immunoblotting procedures. J Immunol Methods 2001; 253: 125–31
Casoni I, Ricci G, Ballarin E, et al. Haematological indices of erythropoietin administration in athletes. Int J Sports Med 1993 Aug; 14(6): 307–11
Gareau R, Gagnon MG, Thellend C, et al. Transferrin soluble receptor: a possible probe for detection of erythropoietin abuse by athletes. Horm Metab Res 1994 Jun; 26(6): 311–2
Souillard A, Audran M, Bressolle F, et al. Recombinant human erythropoietin and pharmacodynamic parameters in athletes: interest of blood sampling for doping control. Br J Clin Pharmacol 1996; 42: 355–60
Bressolle F, Audran M, Gareau R, et al. Population pharmacodynamics for monitoring epoetin in athletes. Clin Drug Invest 1997 Sep; 14(3): 233–42
Audran M, Gareau R, Matecki S, et al. Effects of erythropoietin administration in training athletes and possible indirect detection in doping control. Med Sci Sports Exerc 1999; 31(5): 639–45
Parisotto R, Gore CJ, Hahn AG, et al. Reticulocyte parameters as potential discriminators of recombinant human erythropoietin abuse in elite athletes. Int J Sports Med 2000 Oct; 21(7): 471–9
Russell G, Gore CJ, Ashenden MJ, et al. Effects of prolonged low doses of recombinant human erythropoietin during sub-maximal and maximal exercise. Eur J Appl Physiol 2002; 86: 442–6
Parisotto R, Gore CJ, Emslie KR, et al. A novel method utilizing markers of altered erythropoiesis for the detection of recombinant human erythropoietin abuse in athletes. Haematologica 2000 Jun; 85(6): 564–72
Parisotto R, Wu M, Ashenden MJ, et al. Detection of recombinant human erythropoietin abuse in athletes utilizing markers of altered erythropoiesis. Haematologica 2001 Feb; 86(2): 128–37
Magnani M, Corsi D, Bianchi M, et al. Identification of blood erythroid markers useful in revealing erythropoietin abuse in athletes. Blood Cells Mol Dis 2001 May/Jun; 27(3): 559–71
Ashenden MA. A strategy to detect blood doping in sport. Haematologica 2002; 87(3): 225–31
Breymann C. Erythropoietin. Baillieres Clin Endocrinol Metab 2000; 14(1): 135–45
Ekblom B. Blood boosting and sport. Baillieres Clin Endocrinol Metab 2000; 14(1): 89–98
Wilber RL. Detection of DNA-recombinant human epoetin-alpha as a pharmacological ergogenic aid. Sports Med 2002; 32(2): 125–42
Jelkmann W. Use of recombinant human erythropoietin as an antianemic and performance enhancing drug. Curr Pharm Biotech 2000 Jul; 1(1): 11–31
Fried W. The liver as a source of extrarenal erythropoietin production. Blood 1972 Nov; 40(5): 671–7
Bazan JF. A novel family of growth factor receptors: a common binding domain in the growth hormone, prolactin, erythropoietin and IL-6 receptors, and the p75-2 β-chain. Biochem Biophys Res Commun 1989 Oct; 164(2): 788–95
Bren A, Kandus A, Varl J, et al. A comparison between epoetin omega and epoetin alfa in the correction of anemia in hemodialysis patients: a prospective, controlled crossover study. Artif Organs 2002 Feb; 26(2): 91–7
Sikole A, Spasovski G, Zafirov D, et al. Epoetin omega for treatment of anaemia in maintenance hemodialysis patients. Clin Nephrol 2002 Mar; 57(3): 237–45
Korbett SM. Anemia and erythropoietin in hemodialysis and continuous ambulatory peritoneal dialysis. Kidney Int 1993; 43(1): 111–9
Macdougall IC, Gray SJ, Elston O, et al. Pharmacokinetics of novel erythropoiesis stimulating protein compared with epoetin alfa in dialysis. J Am Soc Nephrol 1999 Nov; 10(11): 2392–5
Egrie JC, Browne JK. Development and characterization of novel erythropoiesis stimulating protein (NESP). Nephrol Dial Transplant 2001; 16Suppl. 3: 3–13
Ibbotson T, Goa KL. Darbepoietin. Drugs 2001; 61(14): 2097–104
De Bree F. Genomic-based drugs in R&D: the promise of a new era [online]. Genomics-based drugs data report and regenerative therapy 2001; 1(1): 5–19. Available from URL: www.p-rous.com/journals/gddr/sample/html/gddr010005/gd-dr010005.html [Accessed 2002 Sep 10]
Swiss National Science Fondation Adieu la Seringue, voici les implants [online]. Available from URL: www.snf.ch/fr/com/prr/prr_cur_mar_19.asp [Accessed 2003 Jan 8]
Maitani Y, Moriya H, Shimoda N, et al. Distribution characteristics of entrapped recombinant human erythropoietin in liposomes and its intestinal absorption in rats. Int J Pharm 1999 Aug; 185: 13–22
Wells JA. Hormone mimicry. Science 1996 Jul; 273(5274): 449–50
Kessler P, Podsakoff G, Chen X, et al. Gene delivery to skeletal muscle results in sustained expression and systemic delivery of a therapeutic protein. Proc Natl Acad Sci U S A 1996; 93: 14082–7
Zhou S, Murphy JE, Escobedo JA, et al. Adeno-associated virus-mediated delivery of erythropoietin leads to sustained elevation of hematocrit in nonhuman primates. Gene Ther 1998; 5: 665–70
Adam D. Gene therapy may be up to speed for cheats at 2008 Olympics. Nature 2001 Dec 6; 414: 569–70
Friedmann T, Koss JO. Gene transfer and athletics: an impending problem. Mol Ther 2001 Jun; 3(6): 819–20
Koury MJ, Bondurant MC. The molecular mechanism of erythropoietin action. Eur J Biochem 1992 Dec; 210(3): 649–63
Cotes PM, Brozovic B. Diurnal variation of serum immunoreactive erythropoietin in a normal subject. Clin Endocrinol 1982 Oct; 17(4): 419–22
Miller ME, Garcia JF, Cohen RA, et al. Diurnal levels of immunoreactive erythropoietin in normal subjects and subjects with chronic lung disease. Br J Haematol 1981 Oct; 49(2): 189–200
Ekblom B. Blood doping and erythropoietin: the effects of variation in haemoglobin concentration and other related factors on physical performance. Am J Sports Med 1996; 24Suppl. 6: S40–S42
Raine AE. Hypertension, blood viscosity, and cardiovascular morbidity in renal failure: implications of erythropoietin therapy. Lancet 1988 Jan 16; I(8577): 97–100
Berglund B, Ekblom B. Effect of recombinant human erythropoietin treatment on blood pressure and some haematological parameters in healthy men. J Int Med 1991; 229: 125–30
Carlini RG, Reyes AA, Rothstein M. Recombinant human erythropoietin stimulates angiogenesis in vitro. Kidney Int 1995; 47: 740–5
Maschio G. Erythropoietin and systemic hypertension. Nephrol Dial Transplant 1995; 10Suppl. 2: 74–9
Cazzola M. A global strategy for prevention and detection of blood doping with erythropoietin and related drugs. Haematologica 2000; 85(6): 561–3
Piron M, Loo M, Gothot A, et al. Cessation of intensive treatment with recombinant human erythropoietin is followed by secondary anemia. Blood 2001 Jan 15; 97(2): 442–8
Casadevall N, Nataf J, Viron B, et al. Pure red-cell aplasia and antierythropoietin antibodies in patients treated with recombinant erythropoietin. N Engl J Med 2002; 346(7): 469–75
Cazzola M. Further concerns about the medical risks of blood doping [editorial]. Haematologica 2002; 87(3): 232
Ekblom B, Berglund B. Effect of erythropoietin administration on maximal aerobic power in man. Scand J Med Sci Sports 1991; 1: 88–93
Birkeland K, Hemmersbach P. The future of doping control in athletes: issues related to blood sampling. Sports Med 1999; 28: 25–30
Birkeland K, Stray-Gundersen J, Hemmersbach P, et al. Effect of rhEPO administration on serum levels of sTfR and cycling performance. Med Sci Sports Exerc 2000 Jul; 32(7): 1238–43
McMahon FG, Vargas R, Ryan M, et al. Pharmacokinetics and effects of recombinant human erythropoietin after intravenous and subcutaneous injection in healthy volunteers. Blood 1990; 76(9): 1718–22
Jensen JD, Jensen LW, Madsen JK. The pharmacokinetics of recombinant human erythropoietin after subcutaneous injection at different sites. Eur J Clin Pharmacol 1994; 46: 333–7
Jensen JD, Madsen JK, Jensen LW, et al. Reduced production, absorption and elimination erythropoietin in uremia compared with healthy volunteers. J Am Soc Nephrol 1994; 5: 177–85
Jensen JD, Jensen LW, Madsen JK, et al. The metabolism of erythropoietin in liver cirrhosis patients compared with healthy volunteers. Eur J Haematol 1995; 54: 111–6
Bressolle F, Audran M, Gareau R, et al. Comparison of a direct and indirect population pharmacodynamic model: application to recombinant human erythropoietin in athletes. J Pharmacokinet Biopharm 1997; 25: 263–75
Flaharty K, Caro J, Erslev A, et al. Pharmacokinetics and erythropoietic response to human recombinant erythropoietin in healthy men. Clin Pharmacol Ther 1990 May; 47(5): 557–64
Halstenson CE, Macres M, Katz SA, et al. Comparative pharmacokinetics and pharmacodynamics of epoetin alpha and epoetin beta. Clin Pharmacol Ther 1991; 50: 702–12
Cheung WK, Goon BL, Giulfoyle MC, et al. Pharmacokinetics and pharmacodynamics of recombinant erythropoietin after single and multiple subcutaneous doses to healthy subjects. Clin Pharmacol Ther 1998; 64(4): 412–23
Cheung WK, Natarajan J, Sanders M, et al. Comparative pharmacokinetics, safety, and tolerability after subcutaneous administration of recombinant human erythropoietin formulated with different stabilizers. Biopharm Drug Dispos 2000; 21: 211–9
Gaudard A, Varlet-Marie E, Audran M, et al. Pharmacokinetic-Pharmacodynamic modelling of recombinant human erythropoietin in athletes: a population approach. Clin Drug Invest. In press
Markham A, Bryson HM. Epoetin alpha: a review of its pharmacodynamic and pharmacokinetic properties and therapeutic use in nonrenal applications. Drugs 1995 Feb; 49(2): 232–54
Bommer J, Barth HP, Zeierand M. Efficacy comparison of intravenous and subcutaneous recombinant human erythropoietin administration in hemodialysis patients. Contrib Nephr 1991; 88: 136–43
Klausen T, Dela F, Hippe E, et al. Diurnal variations of serum erythropoietin in trained and untrained subjects. Eur J Appl Physiol 1993; 67: 545–8
Schmidt W, Eckardt KU, Hilgendorf A, et al. Effect of maximal and submaximal exercise under normoxic and hypoxic conditions on serum erythropoietin level. Int J Sports Med 1991; 12: 457–61
Schwandt HJ, Heyduck B, Genga HC, et al. Influence of prolonged physical exercise on the erythropoietin concentration in blood. Eur J Appl Physiol 1991; 63: 463–6
Veng-Pedersen P, Widness JA, Pereira LM, et al. Kinetic evaluation of nonlinear drug elimination by a disposition decomposition analysis: application to the analysis of the linear elimination kinetics of erythropoietin in adult humans. J Pharm Sci 1995 Jun; 84(6): 760–7
Salmonson T, Danielson BG, Wikström B. The pharmacokinetics of recombinant erythropoietin after intravenous and subcutaneous administration to healthy subjects. Br J Clin Pharmacol 1990; 29: 709–13
Tsunoo M. Phase I study of KRN5702 (epoetin alfa) subcutaneous administration. Rinsho Iyaku 1992; 8(11): 2549–64
Sans T, Joven J, Vilella E, et al. Pharmacokinetics of several subcutaneous doses of erythropoietin: potential implications for blood transfusion. Clin Exp Pharmacol Physiol 2000 Mar; 27(3): 179–84
Hayashi N, Kinoshita H, Yukawa E, et al. Pharmacokinetic analysis of subcutaneous erythropoietin administration with non linear mixed effect model including endogenous production. Br J Clin Pharmacol 1998; 46: 11–9
Macdougall IA, Jones JM, Robinson MI, et al. Subcutaneous erythropoietin therapy: comparison of three different sites of injection. Contrib Nephrol 1991; 88: 152–6
Videman T, Lereim I, Hemmingsson P, et al. Changes in hemoglobin values in elite cross-country skiers from 1987 to 1999. Scan J Med Sci Sports 2000; 10: 89–102
Scheen AJ. Doping with erythropoietin or the misuse of therapeutic advances. Rev Med Liege 1998; 53: 499–502
Report from the French Cycling Federation. Workshop on the detection of abnormal activation of erythropoiesis by pharmacological doses of human recombinant EPO. Paris: Rosnysous-Bois, 1999, May 11
Clarey C. In the arena: no bronze, silver or gold. An obvious answer to drug users [online]. Herald Tribune 2002 Mar 22. Available from URL: http://www.iht.com/ihtsearch.php?id=52056&owner=(International%20Herald%20Tribune)&date=00000000000000 [Accessed 2003 Jan 8]
Acknowledgements
The authors have provided no information on sources of funding or on conflicts of interest directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Additional information
Varlet-Marie and Gaudard have equally contributed to this work and should both be considered as first authors
Rights and permissions
About this article
Cite this article
Varlet-Marie, E., Gaudard, A., Audran, M. et al. Pharmacokinetics/Pharmacodynamics of Recombinant Human Erythropoietins in Doping Control. Sports Med 33, 301–315 (2003). https://doi.org/10.2165/00007256-200333040-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00007256-200333040-00004