Abstract
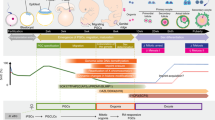
The demonstration of germ cell and haploid gamete development from embryonic stem cells (ESCs) in vitro has engendered a unique set of possibilities for the study of germ cell development and the associated epigenetic phenomenon. The process of embryoid body (EB) differentiation, like teratoma formation, signifies a spontaneous differentiation of ESCs into cells of all three germ layers, and it is from these differentiating aggregates of cells that putative primordial germ cells (PGCs) and more mature gametes can be identified and isolated. The differentiation system presented here requires the differentiation of murine ESCs into EBs and the subsequent isolation of PGCs as well as haploid male gametes from EBs at various stages of differentiation. It serves as a platform for studying the poorly understood process of germ cell allocation, imprint erasure and gamete formation, with 4–6 weeks being required to isolate PGCs as well as haploid cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wylie, C. Germ cells. Cell 96, 165–174 (1999).
Lawson, K.A. & Hage, W.J. Clonal analysis of the origin of primordial germ cells in the mouse. Ciba Found. Symp. 182, 68–84 discussion 84–91 (1994).
Lawson, K.A. et al. Bmp4 is required for the generation of primordial germ cells in the mouse embryo. Genes Dev. 13, 424–436 (1999).
Ying, Y., Qi, X. & Zhao, G.Q. Induction of primordial germ cells from murine epiblasts by synergistic action of BMP4 and BMP8B signaling pathways. Proc. Natl. Acad. Sci. USA 98, 7858–7862 (2001).
Ying, Y. & Zhao, G.Q. Cooperation of endoderm-derived BMP2 and extraembryonic ectoderm-derived BMP4 in primordial germ cell generation in the mouse. Dev. Biol. 232, 484–492 (2001).
Chen, H.-H. & Geijsen, N. Stem Cells in Human Reproduction, Basic Science and Therapeutic Potential Chap. 6 (2006) in the press.
Saitou, M., Barton, S.C. & Surani, M.A. A molecular programme for the specification of germ cell fate in mice. Nature 418, 293–300 (2002).
Vincent, S.D. et al. The zinc finger transcriptional repressor Blimp1/Prdm1 is dispensable for early axis formation but is required for specification of primordial germ cells in the mouse. Development 132, 1315–1325 (2005).
Ohinata, Y. et al. Blimp1 is a critical determinant of the germ cell lineage in mice. Nature 436, 207–213 (2005).
Hubner, K. et al. Derivation of oocytes from mouse embryonic stem cells. Science 300, 1251–1256 (2003).
Toyooka, Y., Tsunekawa, N., Akasu, R. & Noce, T. Embryonic stem cells can form germ cells in vitro . Proc. Natl. Acad. Sci. USA 100, 11457–11462 (2003).
Geijsen, N. et al. Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature 427, 148–154 (2004).
Clark, A.T. et al. Spontaneous differentiation of germ cells from human embryonic stem cells in vitro. Hum. Mol. Genet. 13, 727–739 (2004).
Martin, G.R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl. Acad. Sci. USA 78, 7634–7638 (1981).
Evans, M.J. & Kaufman, M.H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981).
Keller, G.M. In vitro differentiation of embryonic stem cells. Curr. Opin. Cell Biol. 7, 862–869 (1995).
Kehler, J. et al. Oct4 is required for primordial germ cell survival. EMBO Rep. 5, 1078–1083 (2004).
Scholer, H.R., Balling, R., Hatzopoulos, A.K., Suzuki, N. & Gruss, P. Octamer binding proteins confer transcriptional activity in early mouse embryogenesis. EMBO J 8, 2551–2557 (1989).
Scholer, H.R., Hatzopoulos, A.K., Balling, R., Suzuki, N. & Gruss, P. A family of octamer-specific proteins present during mouse embryogenesis: evidence for germline-specific expression of an Oct factor. EMBO J 8, 2543–2550 (1989).
Payer, B. et al. Generation of stella-GFP transgenic mice: a novel tool to study germ cell development. Genesis 44, 75–83 (2006).
MacGregor, G.R., Zambrowicz, B.P. & Soriano, P. Tissue non-specific alkaline phosphatase is expressed in both embryonic and extraembryonic lineages during mouse embryogenesis but is not required for migration of primordial germ cells. Development 121, 1487–1496 (1995).
Fox, N., Damjanov, I., Martinez-Hernandez, A., Knowles, B.B. & Solter, D. Immunohistochemical localization of the early embryonic antigen (SSEA-1) in postimplantation mouse embryos and fetal and adult tissues. Dev. Biol. 83, 391–398 (1981).
Chang, D.H., Angelin-Duclos, C. & Calame, K. BLIMP-1: trigger for differentiation of myeloid lineage. Nat. Immunol. 1, 169–176 (2000).
Koshimizu, U., Watanabe, M. & Nakatsuji, N. Retinoic acid is a potent growth activator of mouse primordial germ cells in vitro . Dev. Biol. 168, 683–685 (1995).
Mizuno, K. et al. Genes associated with the formation of germ cells from embryonic stem cells in cultures containing different glucose concentrations. Mol. Reprod. Dev. 73, 437–445 (2006).
Oishi, K., Barchi, M., Au, A.C., Gelb, B.D. & Diaz, G.A. Male infertility due to germ cell apoptosis in mice lacking the thiamin carrier, Tht1. A new insight into the critical role of thiamin in spermatogenesis. Dev. Biol. 266, 299–309 (2004).
Wong, W.Y., Thomas, C.M., Merkus, J.M., Zielhuis, G.A. & Steegers-Theunissen, R.P. Male factor subfertility: possible causes and the impact of nutritional factors. Fertil. Steril. 73, 435–442 (2000).
Fenderson, B.A., O'Brien, D.A., Millette, C.F. & Eddy, E.M. Stage-specific expression of three cell surface carbohydrate antigens during murine spermatogenesis detected with monoclonal antibodies. Dev. Biol. 103, 117–128 (1984).
Ventela, S. et al. Regulation of acrosome formation in mice expressing green fluorescent protein as a marker. Tissue Cell 32, 501–507 (2000).
Kimmins, S. & Sassone-Corsi, P. Chromatin remodelling and epigenetic features of germ cells. Nature 434, 583–589 (2005).
Nayernia, K. et al. In vitro-differentiated embryonic stem cells give rise to male gametes that can generate offspring mice. Dev. Cell 11, 125–132 (2006).
Novak, I. et al. Mouse embryonic stem cells form follicle-like ovarian structures but do not progress through meiosis. Stem Cells 24, 1931–1936 (2006).
Southern, E. Southern blotting. Nat. Protocols 1, 518–525 (2006).
Biniszkiewicz, D. et al. Dnmt1 overexpression causes genomic hypermethylation, loss of imprinting, and embryonic lethality. Mol. Cell. Biol. 22, 2124–2135 (2002).
Lucifero, D., Mertineit, C., Clarke, H.J., Bestor, T.H. & Trasler, J.M. Methylation dynamics of imprinted genes in mouse germ cells. Genomics 79, 530–538 (2002).
Oulad-Abdelghani, M. et al. Characterization of a premeiotic germ cell-specific cytoplasmic protein encoded by Stra8, a novel retinoic acid-responsive gene. J. Cell Biol. 135, 469–477 (1996).
Lee, J.H., Engel, W. & Nayernia, K. Stem cell protein Piwil2 modulates expression of murine spermatogonial stem cell expressed genes. Mol. Reprod. Dev. 73, 173–179 (2006).
Kubota, H., Avarbock, M.R. & Brinster, R.L. Growth factors essential for self-renewal and expansion of mouse spermatogonial stem cells. Proc. Natl. Acad. Sci. USA 101, 16489–16494 (2004).
Kaneko, K.J. & DePamphilis, M.L. Soggy, a spermatocyte-specific gene, lies 3.8 kb upstream of and antipodal to TEAD-2, a transcription factor expressed at the beginning of mouse development. Nucleic Acids Res. 28, 3982–3990 (2000).
Kohn, M.J., Kaneko, K.J. & DePamphilis, M.L. DkkL1 (Soggy), a Dickkopf family member, localizes to the acrosome during mammalian spermatogenesis. Mol. Reprod. Dev. 71, 516–522 (2005).
Lammers, J.H. et al. The gene encoding a major component of the lateral elements of synaptonemal complexes of the rat is related to X-linked lymphocyte-regulated genes. Mol. Cell. Biol. 14, 1137–1146 (1994).
Chambers, I. et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Mitsui, K. et al. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell 113, 631–642 (2003).
Yamaguchi, S., Kimura, H., Tada, M., Nakatsuji, N. & Tada, T. Nanog expression in mouse germ cell development. Gene Expr. Patterns 5, 639–646 (2005).
Fujiwara, Y. et al. Isolation of a DEAD-family protein gene that encodes a murine homolog of Drosophila vasa and its specific expression in germ cell lineage. Proc. Natl. Acad. Sci. USA 91, 12258–12262 (1994).
Wu, M.H. et al. Sequence and expression of testis-expressed gene 14 (Tex14): a gene encoding a protein kinase preferentially expressed during spermatogenesis. Gene Expr. Patterns 3, 231–236 (2003).
Meuwissen, R.L. et al. A coiled-coil related protein specific for synapsed regions of meiotic prophase chromosomes. EMBO J 11, 5091–5100 (1992).
Yoshida, K. et al. The mouse RecA-like gene Dmc1 is required for homologous chromosome synapsis during meiosis. Mol. Cell 1, 707–718 (1998).
Meistrich, M.L., Mohapatra, B., Shirley, C.R. & Zhao, M. Roles of transition nuclear proteins in spermiogenesis. Chromosoma 111, 483–488 (2003).
Zhao, M. et al. Transition nuclear proteins are required for normal chromatin condensation and functional sperm development. Genesis 38, 200–213 (2004).
Brown, C.R. & Hartree, E.F. Identification of acrosin in mouse spermatozoa. J. Reprod. Fertil. 46, 249–251 (1976).
Kitamura, K., Tanaka, H. & Nishimune, Y. Haprin, a novel haploid germ cell-specific RING finger protein involved in the acrosome reaction. J. Biol. Chem. 278, 44417–44423 (2003).
Eirin-Lopez, J.M., Frehlick, L.J. & Ausio, J. Protamines, in the footsteps of linker histone evolution. J. Biol. Chem. 281, 1–4 (2006).
Wang, P.J., McCarrey, J.R., Yang, F. & Page, D.C. An abundance of X-linked genes expressed in spermatogonia. Nat. Genet. 27, 422–426 (2001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
West, J., Park, IH., Daley, G. et al. In vitro generation of germ cells from murine embryonic stem cells. Nat Protoc 1, 2026–2036 (2006). https://doi.org/10.1038/nprot.2006.303
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.303
This article is cited by
-
Mesenchymal stem cells promote spermatogonial stem/progenitor cell pool and spermatogenesis in neonatal mice in vitro
Scientific Reports (2022)
-
OTOGL, a gelforming mucin protein, is nonessential for male germ cell development and spermatogenesis in mice
Reproductive Biology and Endocrinology (2021)
-
Efficient generation of functional haploid spermatids from human germline stem cells by three-dimensional-induced system
Cell Death & Differentiation (2018)
-
Artificial gametes: perspectives of geneticists, ethicists and representatives of potential users
Medicine, Health Care and Philosophy (2013)
-
Human amniotic epithelial cells differentiate into cells expressing germ cell specific markers when cultured in medium containing serum substitute supplement
Reproductive Biology and Endocrinology (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.